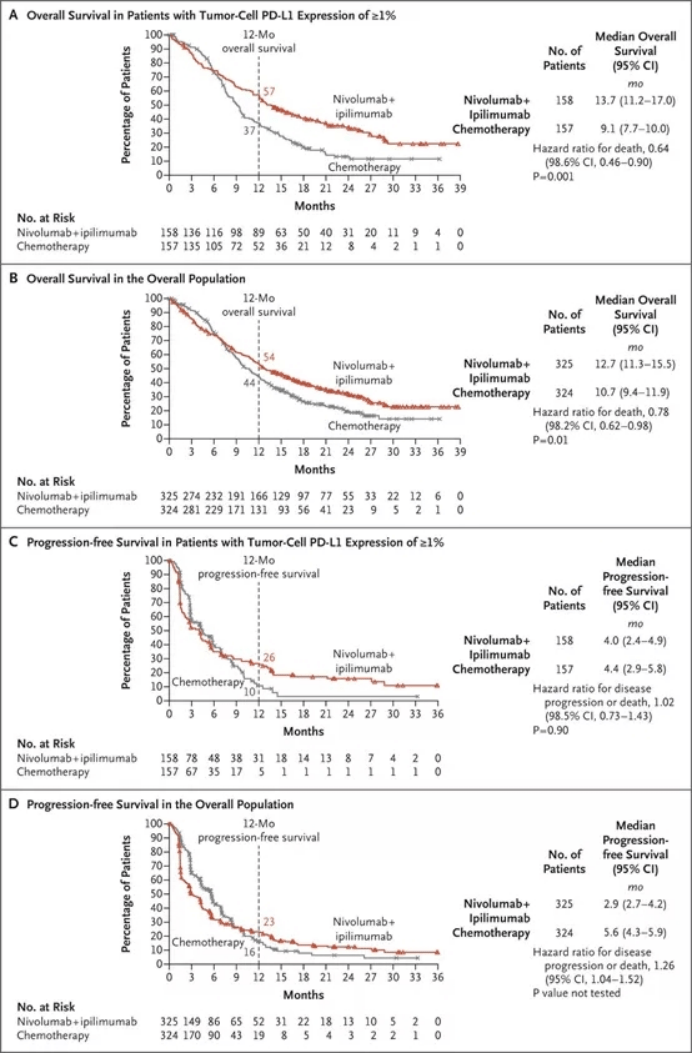 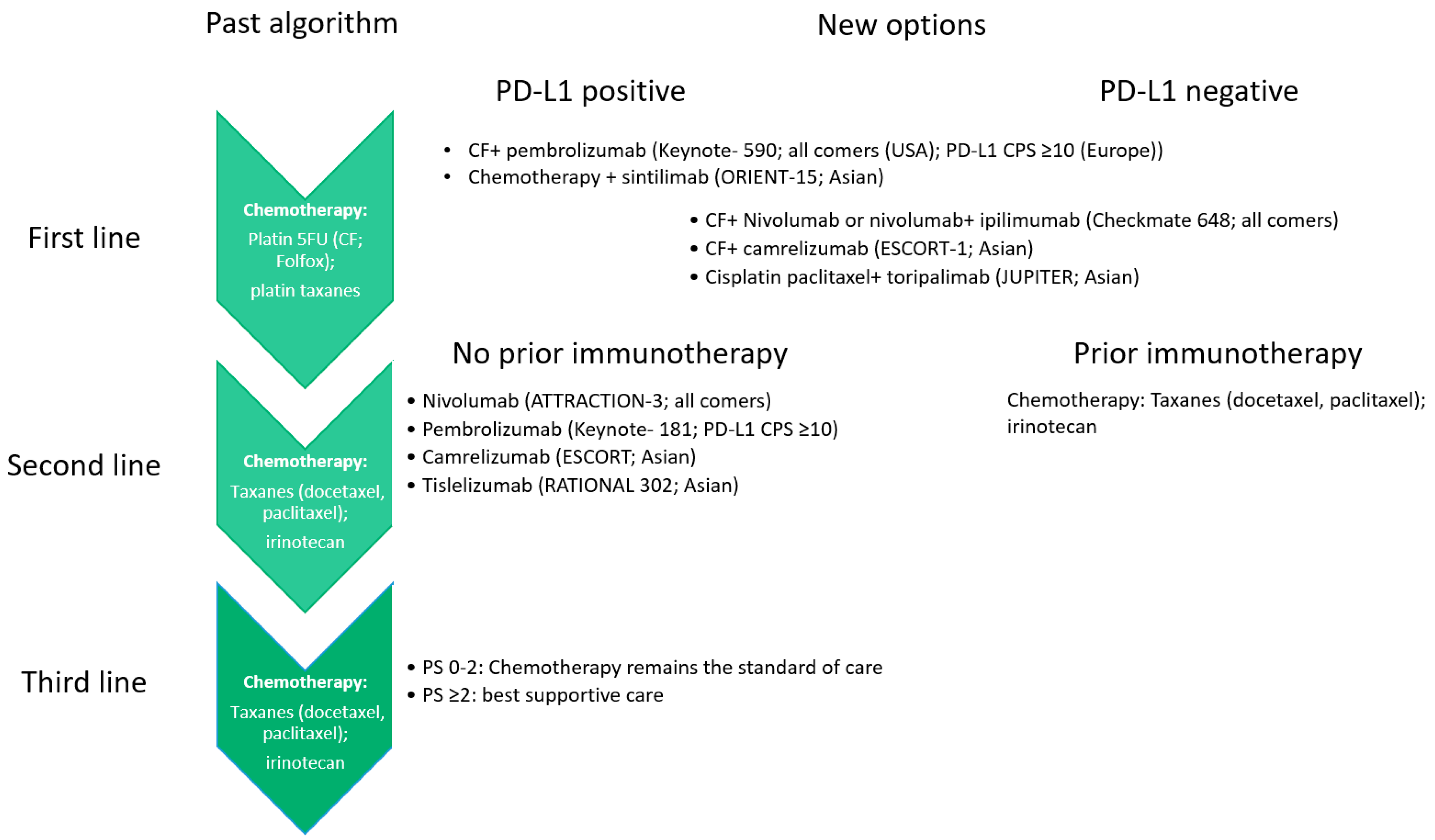
Randomisation was stratified by tumour histology, sex, and PD-L1 expression. Patients were randomly assigned (1:1) by an interactive web response system via permuted blocks (block size of four) to nivolumab (360 mg intravenously every 3 weeks) plus ipilimumab (1 mg/kg intravenously every 6 weeks) combined with histology-based, platinum doublet chemotherapy (intravenously every 3 weeks for two cycles experimental group), or chemotherapy alone (every 3 weeks for four cycles control group). Eligible patients were aged 18 years or older with treatment-naive, histologically confirmed stage IV or recurrent NSCLC, and an Eastern Cooperative Oncology Group performance status of 0-1. This randomised, open-label, phase 3 trial was done at 103 hospitals in 19 countries. We aimed to investigate whether the addition of a limited course (two cycles) of chemotherapy to this combination would further enhance the clinical benefit. 24 Department of Thoracic Oncology, Airway Research Center North, German Center for Lung Research, LungClinic, Grosshansdorf, Germany.įirst-line nivolumab plus ipilimumab has shown improved overall survival in patients with advanced non-small-cell lung cancer (NSCLC).23 Bristol Myers Squibb, Princeton, NJ, USA.22 The Ohio State University Comprehensive Cancer Center, Columbus, OH, USA.21 Austin Hospital, Heidelberg, VIC, Australia.20 Shanghai Lung Cancer Center, Shanghai Chest Hospital, Shanghai JiaoTong University, Shanghai, China.19 Pulmonary and Thoracic Oncology, University of Lille, CHU Lille, INSERM U1189, OncoThAI, Lille, France.18 Léon Bérard Cancer Center, Lyon, France.17 Instituto Alexander Fleming, Buenos Aires, Argentina.16 Barretos Cancer Hospital, Barretos, Brazil.15 Hôpital Lyon Sud, Lyon, Pierre Bénite, France.14 Fundacion Arturo Lopez Perez, Santiago, Metropolitana, Chile.13 Instituto Medico Rio Cuarto SA, Córdoba, Argentina.12 Saitama Cancer Center, Saitama, Japan.11 Institute of Oncology Prof Dr Alexandru Trestioreanu Bucha, Bucharest, Romania.10 Hospital Universitario La Fe, Valencia, Spain.9 Vall d'Hebron University Hospital, Vall d'Hebron Institute of Oncology, Barcelona, Spain.8 Thoracic Oncology Unit, University Hospital of Nantes and INSERM, CRCINA, Nantes, France.7 Instituto Oncológico De Córdoba, Córdoba, Argentina.6 Hospital Nossa Senhora Da Conceição, Porto Alegre, Brazil.5 Ambulatorium Chemioterapii, Bydgoszcz, Poland.4 SF Nectarie Oncology Center, Craiova, Romania.3 Unidad de Gestión Clínica Intercentros de Oncología Médica, Hospitales Universitarios Regional y Virgen de la Victoria, IBIMA, Málaga, Spain.Electronic address: 2 Institutul Oncologic Prof Dr Ion Chiricuta and UMF Iuliu Hatieganu, Cluj-Napoca, Romania. 1 Hospital Universitario 12 de Octubre, CNIO-H12o Lung Cancer Unit, Universidad Complutense & CiberOnc, Madrid, Spain.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
